 
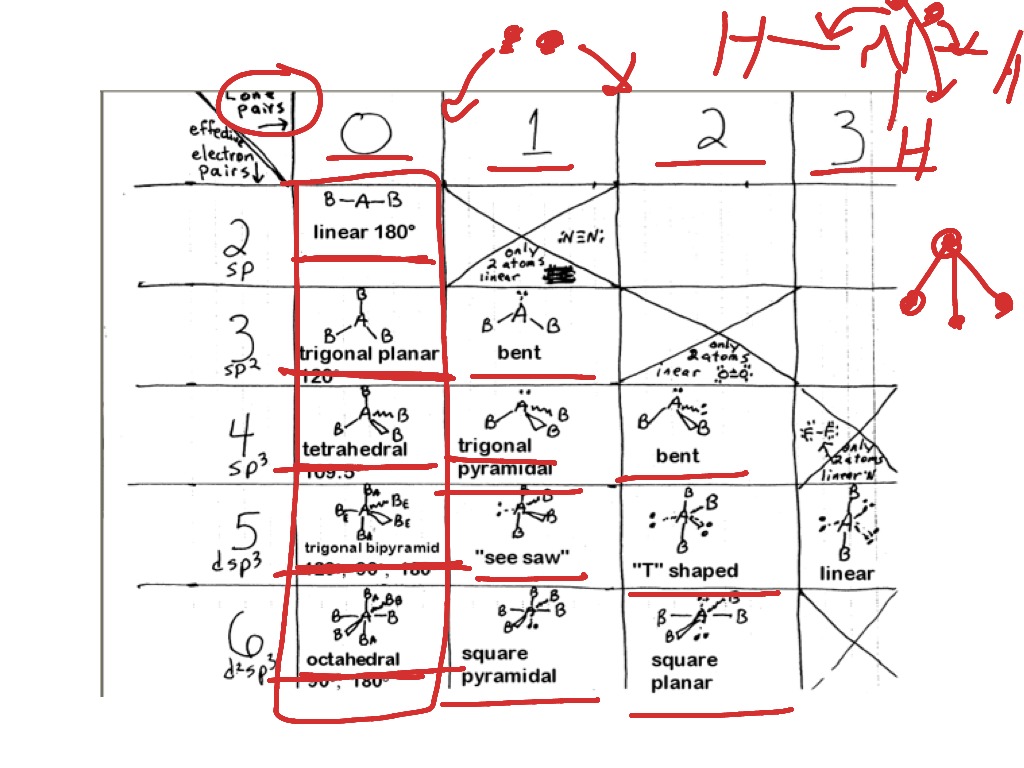
The shapes that are possible are tetrahedral, trigonal planar, trigonal pyramidal, bent, and linear. The valence shell electron pair repulsion (VSEPR) theory is how the geometry of a molecule is determined. Structures on their own, these substances can be used as a starting point for Go deeper into formal charges, or if your students come across these alternative (Those structures and further information onįormal charges are also included at the end of the answer key.) If you wish to Of them violate the octet rule in order to minimize formal charges. A possible extensionĬould be to add more complicated molecules that extend past the octet rule.ĭrawn to satisfy the octet rule, experimental data shows that in reality some Lewis structures can be drawn to satisfy the octet rule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
